Non reducing anomeric carbon1/1/2024  So we've got the one carbon right here, that's this one, and we've got the four carbon over here, so this is again a 1,4 Glycosidic Linkage, but as opposed to Lactose, up here this one is actually Alpha, you can see that this OR Two individual Glocuse units, so we got a Glucose right here and we've got a Glucose right here and they're bound together similarly by a 1,4 Glycoside. And Maltose is, again, a Disaccharide but this time it's made of Now, next step we have Maltose, so let me write that in So we've got the one and a four, and this is a Glycoside Bond, and this one happens toīe in a Beta orientation, so Lactose is a Disaccharide made of Galactose and Glucose joined together by a Beta 1,4 Glycoside Bond. It consists of one Galactose, so this one right here is Galactose, and one Glucose Carbohydrate, and their bound togetherīy a 1,4 Glycoside Bond just like we saw before. True for both human milk and for cow milk, and unlike really most Disaccharides Lactose isn't really appreciably sweet. And Lactose happens to be, really the principalĭisaccharide found in milk and that's actually To save just a bit of time, and what we have here is a Disaccharide, you see two Carbohydrates, two Monosaccharides linked together, and this one happens to be Lactose which you might be familiar with. I'm gonna go and fade in a drawing that I did a little bit ago And if that bit of namingĬonfused you a little bit I went over that in greaterĭetail in a different video, but what I wanna confused on here are some of the common Disaccharides, so let me clear some space, So, in this case, our OR group, which- Our OR group is this whole Carbohydrate is cis with respect to the sixth carbon, so we have a Beta 1,4 Glycosidic Linkage. 
Respect to the sixth carbon it's Beta, and of course if it's trans So same rules apply, if the group is cis with Now the orientation of the OR group on the anomeric carbon. Have further break down our Monosccharides into Alpha and Beta based off the orientation of the anomeric Hydroxide group, we can more specificallyĬall the 1,4 Linkage an Alpha or a Beta Linkage, again, based off what is So we call this a 1,4 Glycosidic Linkage. So, right here would be C4, and it's just the same over here so, right here we have C4, and Here, be right here, just the same, we got aĬ1 of the first Sugar, and the C4 of the second Sugar. Now with Disaccharides, mostĬommonly the Glycosidic Linkage forms between the anomeric carbon, or C1, so remember this is theĪnomeric carbon, the C1, so over in our Glycoside So, two Monosaccharides linked together they're called a Disaccharide. And we have "Di" which means two, and "saccharide" which means sugar, so sugar. Now, when two MonosaccharidesĪre linked together in this fashion by Glycosidic Linkages we call the product a Disaccharide. And it makes sense because what you see with Carbohydrates is that they're choke-full of Hydroxides, they're choke-full of these OH groups, and so, really they canįunction really similarly to Alcohols and reactions, and when this happens, the individual MonosaccharidesĪre linked together to make an Acetal, and we call this linkageĪ Glycosidic Linkage. And I believe that I mentioned that sometimes the AlcoholĬomes in and is reduced, is actually another Carbohydrate. Alright, so in a previous video I talked about how cyclic Monosaccharides like this green cyclic GlucoseĬan react with Alcohols, like this pink Alcohol, to form Acetals and Ketals. therefore, this anomeric carbon can't open up the ring structure and react with the reagent. On the other hand, sucrose's anomeric carbon is not "free" since this carbon is used to link fructose and glucose together. 
Maltose's anomeric carbon is "free" and can therefore open up the ring and reduce the metal ion. This is also the carbon that can open up the ring structure and reduce a metal ion. 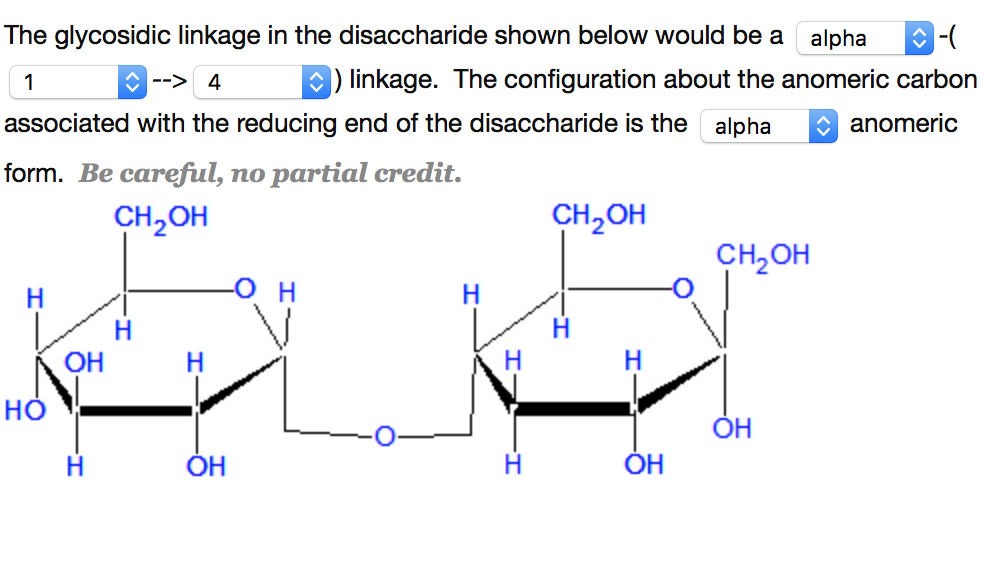
this is the carbon which was hydrolyzed in the straight-chain structure. In the ring structures of sucrose and maltose, you have an anomeric carbon. The reducing ability of disaccharides is defined by the presence of a potential aldehyde or ketone group. Maltose is made up of two glucose units while sucrose is made up of glucose and fructose. Maltose and sucrose are disaccharides, which means that they are made up of two monosaccharides. this means that they are all reducing sugars. Reducing capability is defined by the presence of free or potential aldehyde or ketone group.Īll monosaccharides have free ketone or aldehyde group. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
